A functional view reveals substantial predictability of pollinator-mediated selection
DOI:
https://doi.org/10.26786/1920-7603(2021)673Keywords:
adaptive landscape, causal modelling, path analysis, phenotypic selection, pollinator-mediated selection, selection gradientAbstract
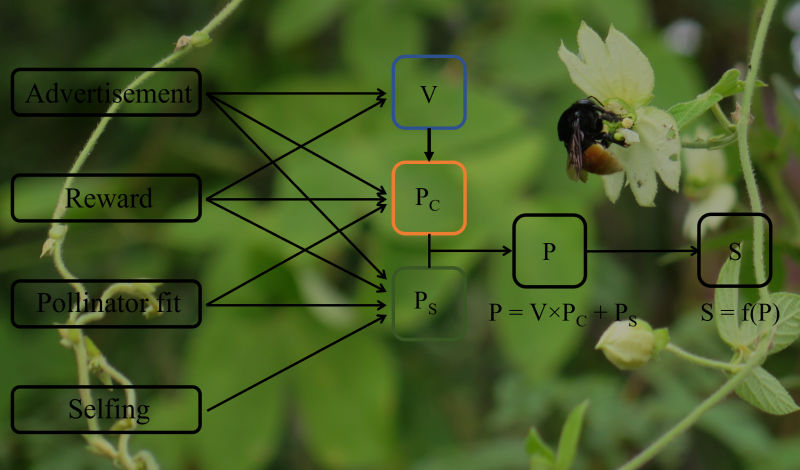
A predictive understanding of adaptation to changing environments hinges on a mechanistic understanding of the extent and causes of variation in natural selection. Estimating variation in selection is difficult due to the complex relationships between phenotypic traits and fitness, and the uncertainty associated with individual selection estimates. Plant-pollinator interactions provide ideal systems for understanding variation in selection and its predictability, because both the selective agents (pollinators) and the process linking phenotypes to fitness (pollination) are generally known. Through examples from the pollination literature, I discuss how explicit consideration of the functional mechanisms underlying trait-performance relationships can clarify the relationship between traits and fitness, and how variation in the ecological context that generates selection can help disentangle biologically important variation in selection from sampling variation. I then evaluate the predictability of variation in pollinator-mediated selection through a survey, reanalysis, and synthesis of results from the literature. The synthesis demonstrates that pollinator-mediated selection often varies substantially among trait functional groups, as well as in time and space. Covariance between patterns of selection and ecological variables provides additional support for the biological importance of observed selection, but the detection of such covariance depends on careful choice of relevant predictor variables as well as consideration of quantitative measurements and their meaning, an aspect often neglected in selection studies.
References
Aigner PA (2005) Variation in pollination performance gradients in a Dudleya species complex: can generalization promote floral divergence? Functional Ecology 19:681-689. DOI: https://doi.org/10.1111/j.1365-2435.2005.01009.x
Albertsen E, Opedal ØH, Bolstad GH, Perez-Barrales R, Hansen T, Pélabon C, Armbruster WS (2021) Using ecological context to interpret spatiotemporal variation in natural selection. Evolution 75:294-309. DOI: https://doi.org/10.1111/evo.14136
Alexandersson R, Johnson SD (2002) Pollinator-mediated selection on flower-tube length in a hawkmoth-pollinated Gladiolus (Iridaceae). Proceedings of the Royal Society B-Biological Sciences 269:631-636. DOI: https://doi.org/10.1098/rspb.2001.1928
Anderson B, Johnson SD (2009) Geographical covariation and local convergence of flower depth in a guild of fly-pollinated plants. New Phytologist 182:533-540. DOI: https://doi.org/10.1111/j.1469-8137.2009.02764.x
Anderson B, Terblanche JS, Ellis AG (2010) Predictable patterns of trait mismatches between interacting plants and insects. BMC Evolutionary Biology 10:204. DOI: https://doi.org/10.1186/1471-2148-10-204
Andersson S (1996) Floral display and pollination success in Senecio jacobaea (Asteraceae): interactive effects of head and corymb size. American Journal of Botany 83:71-75. DOI: https://doi.org/10.1002/j.1537-2197.1996.tb13876.x
Armbruster WS (1988) Multilevel comparative analysis of the morphology, function, and evolution of Dalechampia blossoms. Ecology 69:1746-1761. DOI: https://doi.org/10.2307/1941153
Armbruster WS (1990) Estimating and testing the shapes of adaptive surfaces - the morphology and pollination of Dalechampia blossoms. American Naturalist 135:14-31. DOI: https://doi.org/10.1086/285029
Armbruster WS, Antonsen L, Pélabon C (2005) Phenotypic selection on Dalechampia blossoms: honest signaling affects pollination success. Ecology 86:3323-3333. DOI: https://doi.org/10.1890/04-1873
Armbruster WS, Gong Y-B, Huang S-Q (2011) Are pollination “syndromes” predictive? Asian Dalechampia fit neotropical models. American Naturalist 178:135-143. DOI: https://doi.org/10.1086/660279
Arnold SJ (1983) Morphology, performance and fitness. American Zoologist 23:347-361. DOI: https://doi.org/10.1093/icb/23.2.347
Arnold SJ, Wade MJ (1984a) On the measurement of natural and sexual selection: applications. Evolution 38:720-734. DOI: https://doi.org/10.1111/j.1558-5646.1984.tb00345.x
Arnold SJ, Wade MJ (1984b) On the measurement of natural and sexual selection: theory. Evolution 38:709-719. DOI: https://doi.org/10.1111/j.1558-5646.1984.tb00344.x
Ashman TL, Morgan MT (2004) Explaining phenotypic selection on plant attractive characters: male function, gender balance or ecological context? Proceedings of the Royal Society B-Biological Sciences 271:553-559. DOI: https://doi.org/10.1098/rspb.2003.2642
Bartkowska MP, Johnston MO (2012) Pollinators cause stronger selection than herbivores on floral traits in Lobelia cardinalis (Lobeliaceae). New Phytologist 193:1039-1048. DOI: https://doi.org/10.1111/j.1469-8137.2011.04013.x
Bartkowska MP, Johnston MO (2015) Pollen limitation and its influence on natural selection through seed set. Journal of Evolutionary Biology 28:2097-2105. DOI: https://doi.org/10.1111/jeb.12741
Benkman CW (2013) Biotic interaction strength and the intensity of selection. Ecology Letters 16:1054-1060. DOI: https://doi.org/10.1111/ele.12138
Boberg E, Alexandersson R, Jonsson M, Maad J, Agren J, Nilsson LA (2014) Pollinator shifts and the evolution of spur length in the moth-pollinated orchid Platanthera bifolia. Annals of Botany 113:267-275. DOI: https://doi.org/10.1093/aob/mct217
Bodbyl Roels SA, Kelly JK (2011) Rapid evolution caused by pollinator loss in Mimulus guttatus. Evolution 65:2541-2552. DOI: https://doi.org/10.1111/j.1558-5646.2011.01326.x
Bolstad GH, Armbruster WS, Pélabon C, Pérez-Barrales R, Hansen TF (2010) Direct selection at the blossom level on floral reward by pollinators in a natural population of Dalechampia schottii: full-disclosure honesty? New Phytologist 188:370-384. DOI: https://doi.org/10.1111/j.1469-8137.2010.03429.x
Campbell DR (2009) Using phenotypic manipulations to study multivariate selection of floral trait associations. Annals of Botany 103:1557-1566. DOI: https://doi.org/10.1093/aob/mcp032
Campbell DR, Forster M, Bischoff M (2014) Selection of trait combinations through bee and fly visitation to flowers of Polemonium foliosissimum. Journal of Evolutionary Biology 27:325-336. DOI: https://doi.org/10.1111/jeb.12295
Campbell DR, Powers JM (2015) Natural selection on floral morphology can be influenced by climate. Proceedings of the Royal Society B-Biological Sciences 282:20150178. DOI: https://doi.org/10.1098/rspb.2015.0178
Campbell DR, Waser NM, Melendez-Ackerman EJ (1997) Analyzing pollinator-mediated selection in a plant hybrid zone: hummingbird visitation patterns on three spatial scales. American Naturalist 149:295-315. DOI: https://doi.org/10.1086/285991
Campbell DR, Waser NM, Price MV (1996) Mechanisms of hummingbird-mediated selection for flower width in Ipomopsis aggregata. Ecology 77:1463-1472. DOI: https://doi.org/10.2307/2265543
Campbell DR, Waser NM, Price MV, Lynch EA, Mitchell RJ (1991) Components of phenotypic selection: pollen export and flower corolla width in Ipomopsis aggregata. Ecology 45:1458-1467. DOI: https://doi.org/10.1111/j.1558-5646.1991.tb02648.x
Caruso CM, Eisen KE, Martin RA, Sletvold N (2019) A meta-analysis of the agents of selection on floral traits. Evolution 73:4-14. DOI: https://doi.org/10.1111/evo.13639
Caruso CM, Martin RA, Sletvold N, Morrissey MB, Wade MJ, Augustine KE et al. (2017) What are the environmental determinants of phenotypic selection? A meta-analysis of experimental studies. The American Naturalist 190:363-376. DOI: https://doi.org/10.1086/692760
Castellanos MC, Wilson P, Thomson JD (2004) 'Anti-bee' and 'pro-bird' changes during the evolution of hummingbird pollination in Penstemon flowers. Journal of Evolutionary Biology 17:876-885. DOI: https://doi.org/10.1111/j.1420-9101.2004.00729.x
Chapurlat E, Ågren J, Sletvold N (2015) Spatial variation in pollinator-mediated selection on phenology, floral display and spur length in the orchid Gymnadenia conopsea. New Phytologist 208:1264-1275. DOI: https://doi.org/10.1111/nph.13555
Conner JK (1996) Understanding natural selection: an approach integrating selection gradients, multiplicative fitness components, and path analysis. Ethology Ecology & Evolution 8:387-397. DOI: https://doi.org/10.1080/08927014.1996.9522911
Conner JK (2003) Artificial selection: a powerful tool for ecologists. Ecology 84:1650-1660. DOI: https://doi.org/10.1890/0012-9658(2003)084[1650:ASAPTF]2.0.CO;2
Cresswell JE (1998) Stabilizing selection and the structural variability of flowers within species. Annals of Botany 81:463-473. DOI: https://doi.org/10.1006/anbo.1998.0594
Cresswell JE (2000) Manipulation of female architecture in flowers reveals a narrow optimum for pollen deposition. Ecology 81:3244-3249. DOI: https://doi.org/10.1890/0012-9658(2000)081[3244:MOFAIF]2.0.CO;2
Darwin CR (1862) On the various contrivances by which British and foreign orchids are fertilised by insects, and on the good effects of intercrossing. John Murray, London
De Lisle SP, Svensson EI (2017) On the standardization of fitness and traits in comparative studies of phenotypic selection. Evolution 71:2313-2326. DOI: https://doi.org/10.1111/evo.13325
Eisen KE, Wruck AC, Geber MA (2020) Floral density and co‐occurring congeners alter patterns of selection in annual plant communities. Evolution in press. DOI: https://doi.org/10.1111/evo.13960
Ellis AG, Johnson SD (2010) Gender differences in the effects of floral spur length manipulation on fitness in a hermaphrodite orchid. International Journal of Plant Sciences 171:1010-1019. DOI: https://doi.org/10.1086/656351
Emel SL, Franks SJ, Spigler RB (2017) Phenotypic selection varies with pollination intensity across populations of Sabatia angularis. New Phytologist 215:813-824. DOI: https://doi.org/10.1111/nph.14608
Endler JA (1986) Natural selection in the wild. Princeton Univ. Press, Princeton, NJ
Fenster CB, Armbruster WS, Wilson P, Dudash MR, Thomson JD (2004) Pollination syndromes and floral specialization. Annual Review of Ecology Evolution and Systematics 35:375-403. DOI: https://doi.org/10.1146/annurev.ecolsys.34.011802.132347
Gauzere J, Teuf B, Davi H, Chevin LM, Caignard T, Leys B, Delzon S, Ronce O, Chuine I (2020) Where is the optimum? Predicting the variation of selection along climatic gradients and the adaptive value of plasticity. A case study on tree phenology. Evolution Letters 4:109-123. DOI: https://doi.org/10.1002/evl3.160
Gomez JM, Bosch J, Perfectti F, Fernandez JD, Abdelaziz M, Camacho JP (2008) Spatial variation in selection on corolla shape in a generalist plant is promoted by the preference patterns of its local pollinators. Proc Biol Sci 275:2241-2249. DOI: https://doi.org/10.1098/rspb.2008.0512
Gómez JM, Perfectti F, Bosch J, Camacho JPM (2009) A geographic selection mosaic in a generalized plant-pollinator-herbivore system. Ecological Monographs 79:245-263. DOI: https://doi.org/10.1890/08-0511.1
Grant V, Grant KA (1965) Flower pollination in the Phlox family. Columbia University Press, New York
Hansen V-I, Totland Ø (2006) Pollinator visitation, pollen limitation, and selection on flower size through female function in contrasting habitats within a population of Campanula persicifolia. Canadian Journal of Botany 84:412-420. DOI: https://doi.org/10.1139/b06-012
Harder LD, Johnson SD (2009) Darwin's beautiful contrivances: evolutionary and functional evidence for floral adaptation. New Phytologist 183:530-545. DOI: https://doi.org/10.1111/j.1469-8137.2009.02914.x
Hereford J, Hansen TF, Houle D (2004) Comparing strengths of directional selection: how strong is strong? Evolution 58:2133-2143. DOI: https://doi.org/10.1111/j.0014-3820.2004.tb01592.x
Herrera CM, Castellanos MC, Medrano M (2006) Geographical context of floral evolution: towards as improved research programme in floral diversification. In: Harder LD, Barrett SC (eds) Ecology and evolution of flowers. Oxford University Press, Oxford, UK, pp 278-294 DOI: https://doi.org/10.1093/oso/9780198570851.003.0015
Hodgins KA, Barrett SCH (2008) Natural selection on floral traits through male and female function in wild populations of the heterostylous daffodil Narcissus triandrus. Evolution 62:1751-1763. DOI: https://doi.org/10.1111/j.1558-5646.2008.00404.x
Johnson SD, Steiner KE (1997) Long-tongued fly pollination and evolution of floral spur length in the Disa draconis complex (Orchidaceae). Evolution 51:45-53. DOI: https://doi.org/10.1111/j.1558-5646.1997.tb02387.x
Kingsolver JG, Hoekstra HE, Hoekstra JM, Berrigan D, Vignieri SN, Hill CE, Hoang A, Gibert P, Beerli P (2001) The strength of phenotypic selection in natural populations. The American Naturalist 157:245-261. DOI: https://doi.org/10.1086/319193
Kingsolver JG, Schemske DW (1991) Path analyses of selection. Trends in Ecology & Evolution 6:276-280. DOI: https://doi.org/10.1016/0169-5347(91)90004-H
Kulbaba MW, Worley AC (2012) Selection on floral design in Polemonium brandegeei (Polemoniaceae): female and male fitness under hawkmoth pollination. Evolution 66:1344-1359. DOI: https://doi.org/10.1111/j.1558-5646.2011.01536.x
Kulbaba MW, Worley AC (2013) Selection on Polemonium brandegeei (Polemoniaceae) flowers under hummingbird pollination: in opposition, parallel, or independent of selection by hawkmoths? Evolution 67:2194-2206. DOI: https://doi.org/10.1111/evo.12102
La Rosa RJ, Conner JK (2017) Floral function: effects of traits on pollinators, male and female pollination success, and female fitness across three species of milkweeds (Asclepias). American Journal of Botany 104:150-160. DOI: https://doi.org/10.3732/ajb.1600328
Lande R, Arnold SJ (1983) The measurement of selection on correlated characters. Evolution 37:1210-1226. DOI: https://doi.org/10.1111/j.1558-5646.1983.tb00236.x
Maad J (2000) Phenotypic selection in hawkmoth-pollinated Platanthera bifolia: targets and fitness surfaces. Evolution 54:112-123. DOI: https://doi.org/10.1111/j.0014-3820.2000.tb00012.x
MacColl AD (2011) The ecological causes of evolution. Trends in Ecology & Evolution 26:514-522. DOI: https://doi.org/10.1016/j.tree.2011.06.009
Mackin CR, Peña JF, Blanco MA, Balfour NJ, Castellanos MC (2021) Rapid evolution of a floral trait following acquisition of novel pollinators. Journal of Ecology 109:2234-2246. DOI: https://doi.org/10.1111/1365-2745.13636
Matsumura S, Arlinghaus R, Dieckmann U (2012) Standardizing selection strengths to study selection in the wild: a critical comparison and suggestions for the future. Bioscience 62:1039-1054. DOI: https://doi.org/10.1525/bio.2012.62.12.6
Minnaar C, Anderson B (2019) Using quantum dots as pollen labels to track the fates of individual pollen grains. Methods in Ecology and Evolution 10:604-614. DOI: https://doi.org/10.1111/2041-210X.13155
Mitchell RJ (1993) Adaptive significance of Ipomopsis aggregata nectar production: observation and experiment in the field. Evolution 47:25-35. DOI: https://doi.org/10.1111/j.1558-5646.1993.tb01196.x
Mitchell RJ (1994) Effects of floral traits, pollinator visitation, and plant size on Ipomopsis aggregata fruit production. American Naturalist 143:870-889. DOI: https://doi.org/10.1086/285637
Moeller DA, Geber MA (2005) Ecological context of the evolution of self-pollination in Clarkia xantiana: population size, plant communities, and reproductive assurance. Evolution 59:786-799. DOI: https://doi.org/10.1111/j.0014-3820.2005.tb01753.x
Morrissey MB (2016) Meta-analysis of magnitudes, differences and variation in evolutionary parameters. Journal of Evolutionary Biology 29:1882-1904. DOI: https://doi.org/10.1111/jeb.12950
Morrissey MB, Hadfield JD (2012) Directional selection in temporally replicated studies is remarkably consistent. Evolution 66:435-442. DOI: https://doi.org/10.1111/j.1558-5646.2011.01444.x
Muchhala N, Thomson JD (2009) Going to great lengths: selection for long corolla tubes in an extremely specialized bat-flower mutualism. Proc Biol Sci 276:2147-2152. DOI: https://doi.org/10.1098/rspb.2009.0102
Nattero J, Sérsic AN, Cocucci AA (2010) Patterns of contemporary phenotypic selection and flower integration in the hummingbird-pollinated Nicotiana glauca between populations with different flower-pollinator combinations. Oikos 119:852-863. DOI: https://doi.org/10.1111/j.1600-0706.2009.17766.x
Nilsson LA (1988) The evolution of flowers with deep corolla tubes. Nature 334:147-149. DOI: https://doi.org/10.1038/334147a0
Opedal ØH (2019) The evolvability of animal-pollinated flowers: towards predicting adaptation to novel pollinator communities. New Phytologist 221:1128-1135. DOI: https://doi.org/10.1111/nph.15403
Opedal ØH, Bolstad GH, Hansen TF, Armbruster WS, Pélabon C (2017) The evolvability of herkogamy: quantifying the evolutionary potential of a composite trait. Evolution 71:1572-1586. DOI: https://doi.org/10.1111/evo.13258
Parachnowitsch AL, Caruso CM (2008) Predispersal seed herbivores, not pollinators, exert selection on floral traits via female fitness. Ecology 89:1802-1810. DOI: https://doi.org/10.1890/07-0555.1
Parachnowitsch AL, Kessler A (2010) Pollinators exert natural selection on flower size and floral display in Penstemon digitalis. New Phytologist 188:393-402. DOI: https://doi.org/10.1111/j.1469-8137.2010.03410.x
Paudel BR, Shrestha M, Burd M, Adhikari S, Sun YS, Li QJ (2016) Coevolutionary elaboration of pollination-related traits in an alpine ginger (Roscoea purpurea) and a tabanid fly in the Nepalese Himalayas. New Phytologist 211:1402-1411. DOI: https://doi.org/10.1111/nph.13974
Pauw A, Stofberg J, Waterman RJ (2009) Flies and flowers in Darwin's race. Evolution 63:268-279. DOI: https://doi.org/10.1111/j.1558-5646.2008.00547.x
Pérez-Barrales R, Bolstad GH, Pélabon C, Hansen TF, Armbruster WS (2013) Pollinators and seed predators generate conflicting selection on Dalechampia blossoms. Oikos 122:1411-1428. DOI: https://doi.org/10.1111/j.1600-0706.2013.20780.x
Rausher MD (1992) The measurement of selection on quantitative traits: biases due to environmental covariances between traits and fitness. Evolution 46:616-626. DOI: https://doi.org/10.1111/j.1558-5646.1992.tb02070.x
Rymer PD, Johnson SD, Savolainen V (2010) Pollinator behaviour and plant speciation: can assortative mating and disruptive selection maintain distinct floral morphs in sympatry? New Phytologist 188:426-436. DOI: https://doi.org/10.1111/j.1469-8137.2010.03438.x
Sahli HF, Conner JK (2011) Testing for conflicting and nonadditive selection: floral adaptation to multiple pollinators through male and female fitness. Evolution 65:1457-1473. DOI: https://doi.org/10.1111/j.1558-5646.2011.01229.x
Sandring S, Ågren J (2009) Pollinator-mediated selection on floral display and flowering time in the perennial herb Arabidopsis lyrata. Evolution 63:1292-1300. DOI: https://doi.org/10.1111/j.1558-5646.2009.00624.x
Scheiner SM, Mitchell RJ, Callahan HS (2000) Using path analysis to measure natural selection. Journal of Evolutionary Biology 13:423-433. DOI: https://doi.org/10.1046/j.1420-9101.2000.00191.x
Schemske DW, Horvitz CC (1984) Variation among floral visitors in pollination ability: a precondition for mutualism specialization. Science 225:519-521. DOI: https://doi.org/10.1126/science.225.4661.519
Schemske DW, Horvitz CC (1988) Plant-animal interactions and fruit production in a neotropical herb: a path analysis. Ecology 69:1128-1137. DOI: https://doi.org/10.2307/1941267
Schemske DW, Horvitz CC (1989) Temporal variation in selection on a floral character. Evolution 43:461-465. DOI: https://doi.org/10.1111/j.1558-5646.1989.tb04240.x
Shaw RG, Geyer CJ, Wagenius S, Hangelbroek HH, Etterson JR (2008) Unifying life-history analyses for inference of fitness and population growth. American Naturalist 172:E35-47. DOI: https://doi.org/10.1086/588063
Shipley B (2016) Cause and correlation in biology. A user's guide to path analysis, structural equations and causal inference with R. Cambridge University Press, Cambridge DOI: https://doi.org/10.1017/CBO9781139979573
Siepielski AM, DiBattista JD, Carlson SM (2009) It's about time: the temporal dynamics of phenotypic selection in the wild. Ecology Letters 12:1261-1276. DOI: https://doi.org/10.1111/j.1461-0248.2009.01381.x
Siepielski AM, Gotanda KM, Morrissey MB, Diamond SE, DiBattista JD, Carlson SM (2013) The spatial patterns of directional phenotypic selection. Ecology Letters 16:1382-1392. DOI: https://doi.org/10.1111/ele.12174
Siepielski AM, Morrissey MB, Buoro M, Carlson SM, Caruso CM, Clegg SM et al. (2017) Precipitation drives global variation in natural selection. Science 355:959-962. DOI: https://doi.org/10.1126/science.aag2773
Sletvold N (2019) The context dependence of pollinator-mediated selection in natural populations. International Journal of Plant Sciences 180:934-943. DOI: https://doi.org/10.1086/705584
Sletvold N, Ågren J (2010) Pollinator-mediated selection on floral display and spur length in the orchid Gymnadenia conopsea. International Journal of Plant Sciences 171:999-1009. DOI: https://doi.org/10.1086/656597
Sletvold N, Ågren J (2011) Nonadditive effects of floral display and spur length on reproductive success in a deceptive orchid. Ecology 92:2167-2174. DOI: https://doi.org/10.1890/11-0791.1
Sletvold N, Ågren J (2014) There is more to pollinator-mediated selection than pollen limitation. Evolution 68:1907-1918. DOI: https://doi.org/10.1111/evo.12405
Sletvold N, Ågren J (2016) Experimental reduction in interaction intensity strongly affects biotic selection. Ecology 97:3091-3098. DOI: https://doi.org/10.1002/ecy.1554
Soteras F, Rubini Pisano MA, Bariles JB, More M, Cocucci AA (2020) Phenotypic selection mosaic for flower length influenced by geographically varying hawkmoth pollinator proboscis length and abiotic environment. New Phytologist 225:985-998. DOI: https://doi.org/10.1111/nph.16192
Stanton M, Young HJ, Ellstrand NC, Clegg JM (1991) Consequences of floral variation for male and female reproduction in experimental populations of wild radish, Raphanus sativus L. Evolution 45:268-280. DOI: https://doi.org/10.1111/j.1558-5646.1991.tb04402.x
Stebbins GL (1974) Flowering plants: evolution above the species level. Belknap Press, Cambridge, MA. DOI: https://doi.org/10.4159/harvard.9780674864856
Steiner KE, Whitehead VB (1990) Pollinator adaptation to oil-secreting flowers - Rediviva and Diascia. Evolution 44:1701-1707. DOI: https://doi.org/10.1111/j.1558-5646.1990.tb03857.x
Temeles EJ, Bishop GA (2019) A hurricane alters pollinator relationships and natural selection on an introduced island plant. Biotropica 51:129-138. DOI: https://doi.org/10.1111/btp.12634
Thompson JN (2005) The geographic mosaic of coevolution. The University of Chicago Press, Chicago, US. DOI: https://doi.org/10.7208/chicago/9780226118697.001.0001
Totland Ø, Andersen HL, Bjelland T, Dahl V, Eide W, Houge S et al. (1998) Variation in pollen limitation among plants and phenotypic selection on floral traits in an early-spring flowering herb. Oikos 82. DOI: https://doi.org/10.2307/3546370
Trunschke J, Sletvold N, Ågren J (2017) Interaction intensity and pollinator-mediated selection. New Phytologist 214:1381-1389. DOI: https://doi.org/10.1111/nph.14479
van Tienderen PH (2000) Elasticities and the link between demographic and evolutionary dynamics. Ecology 81:666-679. DOI: https://doi.org/10.1890/0012-9658(2000)081[0666:EATLBD]2.0.CO;2
Vanhoenacker D, Ågren J, Ehrlen J (2013) Non-linear relationship between intensity of plant-animal interactions and selection strength. Ecology Letters 16:198-205. DOI: https://doi.org/10.1111/ele.12029
Wade MJ, Kalisz S (1990) The causes of natural selection. Evolution 44:1947-1955. DOI: https://doi.org/10.1111/j.1558-5646.1990.tb04301.x
Walker JA (2007) A general model of functional constraints on phenotypic evolution. American Naturalist 170:681-689. DOI: https://doi.org/10.1086/521957
Week B, Nuismer SL (2019) The measurement of coevolution in the wild. Ecology Letters 22:717-725. DOI: https://doi.org/10.1111/ele.13231
Wilson P (1995a) Selection for pollination success and the mechanical fit of Impatiens flowers around bumblebee bodies. Biological Journal of the Linnean Society 55:355-383. DOI: https://doi.org/10.1006/bijl.1995.0047
Wilson P (1995b) Variation in the intensity of pollination in Drosera tracyi: selection is strongest when resources are intermediate. Evolutionary Ecology 9:382-396. DOI: https://doi.org/10.1007/BF01237761
Downloads
Additional Files
Published
How to Cite
Issue
Section
License
Copyright (c) 2021 Øystein Opedal

This work is licensed under a Creative Commons Attribution 4.0 International License.












